|
Dr. Pimchai Chaiyen's Laboratory
: Current systems |
Flavin-dependent enzymes
: OXYGENASE (2)
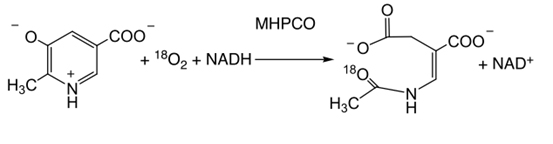
2) 2-methyl-3-hydroxypyridine-carboxylate monooxygenase (MHPCO)
MHPCO catalyzes conversion of 2-methyl-3-hydroxypyridine carboxylate into alpha-(N-acetylaminomethylene)-succinic acid. This reaction is interesting in that MHPCO first catalyzes ortho-hydroxylation of a hydroxy-pyridine compound which is followed a ring-cleavage reaction.
This reaction is unique and among only a few enzymes that can use organic cofactor to catalyze aromatic ring-cleavage reactions.
Current project:
-Identification of residues important for ring-cleavage reactions.
-Engineering of MHPCO for its application as a biocatalyst
Investigator:
Jeerus Sucharitakul
Thikumporn Luanloet

3) 3-hydroxybenzoate 6-hydroxylase
3-hydroxybenzoate 6-hydroxylase (3HB6H) is an enzyme that catalyzes the NADH-dependent conversion of 3-hydroxybenzoate to gentisate. This enzyme is involved in bacterial degradation of aromatic compounds. 3HB6H is useful in chemical synthesis as a biocatalyst to specifically incorporate a hydroxyl group into a phenolic compound at the para-position.
Current project
-Reaction mechanisms of 3HB6H
-Factors controlling active site specificity of 3HB6H
Investigator:
Jeerus Sucharitakul
Dheeradhach Medhanavyn
Tippanate Jongjitsatitmun

4) Bacterial Luciferase
Light-emitting systems are abundant in a wide range of organisms including insects, coelenterate, algae, fungi, and bacteria. The reaction commonly involves the oxygenation of luciferin (substrate) catalyzed by the luciferase (enzyme). For a bacterial luciferase, the overall reaction can be described as follows.
We isolated a new bacterial luciferase from Vibrio campbellii and showed that this enzyme is significantly more thermostable than other bacterial luciferases (Suadee et al., 2007). In addition, we demonstrated for the first time that a LuxG protein encoded in the same operon as the luciferase is a flavin reductase which provides reduced FMN as a substrate for the luciferase reaction (Nijvipakul, et al., 2008). The reduction kinetics of LuxG has been elucidated and shown to be involved with half-sites reactivity (Nijvipakul et al., 2010). Recently, we have constructed a reporter gene based on the bacterial luciferase system (Tinikul et al., 2012)
Current research project:
-Development of luciferase from V. campbellii as gene reporters for bacteria and yeast
Investigators:
Ruchanok Cheotacha
Jittima Phonbuppha

[ ...
to continue ... ]
|